Types of Reaction
There are 5 types of reactions that needs to be recognized:
Synthesis, Decomposition , Single replacement, Double replacement, Combustion.
The 7 diatomic elements
There are 7 diatomic elements:
I2, H2, N2, Br2, O2, Cl2, F2
Balancing Equation Rules
1. Determine correct formula and physical states.
2. Write equation with reactants on left and products on rights.
3. Count the number of atoms of each elements in reactants and products.
4. Balance equation using coefficients. Never change subscripts!
5.Check atom or polyatomic ion to be sure equation is balanced.
6. Make sure coefficients are in the lowest possible.
Always balance the chemical equations.
Symbols:
(s)=solid
(l)=liquid
(g)=gas
(aq)=dissolved in water
Sig Fig
The number of sig figs is the number of digits by doing the measures. It also includes one estimated digit. It can be precise or accurate numbers
Rules for Working with Significant Figures:
There are 5 types of reactions that needs to be recognized:
Synthesis, Decomposition , Single replacement, Double replacement, Combustion.
The 7 diatomic elements
There are 7 diatomic elements:
I2, H2, N2, Br2, O2, Cl2, F2
Balancing Equation Rules
1. Determine correct formula and physical states.
2. Write equation with reactants on left and products on rights.
3. Count the number of atoms of each elements in reactants and products.
4. Balance equation using coefficients. Never change subscripts!
5.Check atom or polyatomic ion to be sure equation is balanced.
6. Make sure coefficients are in the lowest possible.
Always balance the chemical equations.
Symbols:
(s)=solid
(l)=liquid
(g)=gas
(aq)=dissolved in water
Sig Fig
The number of sig figs is the number of digits by doing the measures. It also includes one estimated digit. It can be precise or accurate numbers
Rules for Working with Significant Figures:
- Leading zeros are never significant.
Embedded zeros are always significant.
Trailing zeros are significant only if the decimal point is specified.
Hint: Change the number to scientific notation. It is easier to see. - Addition or Subtraction:
The last digit retained is set by the first doubtful digit. - Multiplication or Division:
The answer contains no more significant figures than the least accurately known number.
"601_ChemRxn.htm." 601_ChemRxn.htm. N.p., n.d. Web. 28 May 2014.
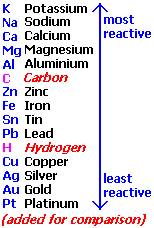
"Gcse Reactivity Series of Metals, Metallic Activity Order Word/symbol Equations of Reactions of Metals with Air/oxygen, Water, Hydrochloric Acid, Sulphuric/sulfuric Acid, Nitric Acid Igcse KS4 Science Chemistry Revision Notes Revising." Gcse Reactivity Series of Metals, Metallic Activity Order Word/symbol Equations of Reactions of Metals with Air/oxygen, Water, Hydrochloric Acid, Sulphuric/sulfuric Acid, Nitric Acid Igcse KS4 Science Chemistry Revision Notes Revising. N.p., n.d. Web. 28 May 2014.
"4.2 Precipitation Reactions." Chemistry: The Central Science, Chapter 4, Section 2. N.p., n.d. Web. 28 May 2014.
"Gcse Reactivity Series of Metals, Metallic Activity Order Word/symbol Equations of Reactions of Metals with Air/oxygen, Water, Hydrochloric Acid, Sulphuric/sulfuric Acid, Nitric Acid Igcse KS4 Science Chemistry Revision Notes Revising." Gcse Reactivity Series of Metals, Metallic Activity Order Word/symbol Equations of Reactions of Metals with Air/oxygen, Water, Hydrochloric Acid, Sulphuric/sulfuric Acid, Nitric Acid Igcse KS4 Science Chemistry Revision Notes Revising. N.p., n.d. Web. 28 May 2014.
"4.2 Precipitation Reactions." Chemistry: The Central Science, Chapter 4, Section 2. N.p., n.d. Web. 28 May 2014.